Publications
Cover Images
2025
- Palacios, P. M.; Li, X.; Rifayee, S. B. J. S.; Tang, H.; Karabencheva-Christova, T.; Christov, C. Z.; Chang, W.-C.; Guo, Y. Regio- and Stereo-Selective Halogenation by an Iron(II)- and 2-Oxoglutarate-Dependent Halogenase in the Biosynthesis of Halogenated Nucleosides. J. Am. Chem. Soc. 2025, 147, 52, 48166–48179. https://doi.org/10.1021/jacs.5c16374.
- Cherilakkudy, F. H.; Thomas, M. G.; Varghese, A.; Waheed, S. O.; Krishnan, A.; Venditti, V.; Schofield, C. J.; Li, D.; Christov, C. Z.; Karabencheva-Christova, T. G. Revealing the Catalytic Mechanism of the Fe(II)/2-Oxoglutarate-Dependent Human Epigenetic Modifying Enzyme ALKBH5. Cell Rep. Phys. Sci. 2025, 6 (8), 102779. https://doi.org/10.1016/j.xcrp.2025.102779.
- Melayikandy, S.; Krishnan, A.; Varghese, A.; Jaber Sathik Rifayee, S. B.; Waheed, S. O.; Ramanan, R.; Li, D.; Christov, C. Z.; Karabencheva-Christova, T. G. Catalytic Mechanism of the Bacterial Non-Heme Fe(II) and 2-Oxoglutarate Dependent Enzyme AlkB with Single-Stranded DNA Containing Complex Guanine Adducts. Inorg. Chem. 2025, 64 (30), 15650–15666. https://doi.org/10.1021/acs.inorgchem.5c02176.
- Chatterjee, S.; Rankin, J. A.; Farrugia, M. A.; Rifayee, S. B. J. S.; Christov, C. Z.; Hu, J.; Hausinger, R. P. Biochemical, Structural, and Conformational Characterization of a Fungal Ethylene-Forming Enzyme. Biochemistry 2025, 64 (9), 2054–2067. https://doi.org/10.1021/acs.biochem.5c00038.
- Jaber Sathik Rifayee, S. B.; Thomas, M. G.; Christov, C. Z. Revealing the nature of the second branch point in the catalytic mechanism of the Fe(ii)/2OG-dependent ethylene forming enzyme RSC Chem. Sci., 2025,16, 7667-7684. https://doi.org/10.1039/D4SC08378D.
2024
- Thomas, M. G.; Jaber Sathik Rifayee, S. B.; Christov, C. Z. How Do Variants of Residues in the First Coordination Sphere, Second Coordination Sphere, and Remote Areas Influence the Catalytic Mechanism of Non-Heme Fe(II)/2-Oxoglutarate Dependent Ethylene-Forming Enzyme? ACS Catal. 2024, 14, 24, 18550–18569. https://doi.org/10.1021/acscatal.4c04010.
- Chatterjee, S.; Fellner, M.; Rankin, JoelA.; Thomas, M. G.; J S Rifayee, S. B.; Christov, C. Z.; Hu, J.; Hausinger, R. P. Structural, Spectroscopic, and Computational Insights from Canavanine-Bound and Two Catalytically Compromised Variants of the Ethylene-Forming Enzyme. Biochemistry 2024, 63 (8), 1038–1050. https://doi.org/10.1021/acs.biochem.4c00031.
- Devadas, S.; Thomas, M. G.; Rifayee, S. B. J. S.; Varada, B.; White, W.; Sommer, E.; Campbell, K.; Schofield, C. J.; Christov, C. Origins of Catalysis in Non‐Heme Fe(II)/2‐Oxoglutarate‐Dependent Histone Lysine Demethylase KDM4A with Differently Methylated Histone H3 Peptides. Chem. – Eur. J. 2024, e202403989. https://doi.org/10.1002/chem.202403989.
- Thomas, M. G.; Jaber Sathik Rifayee, S. B.; Chaturvedi, S. S.; Gorantla, K. R.; White, W.; Wildey, J.; Schofield, C. J.; Christov, C. Z. The Unique Role of the Second Coordination Sphere to Unlock and Control Catalysis in Nonheme Fe(II)/2-Oxoglutarate Histone Demethylase KDM2A. Inorg. Chem.2024, 63 (23), 10737–10755. https://doi.org/10.1021/acs.inorgchem.4c01365.
2023
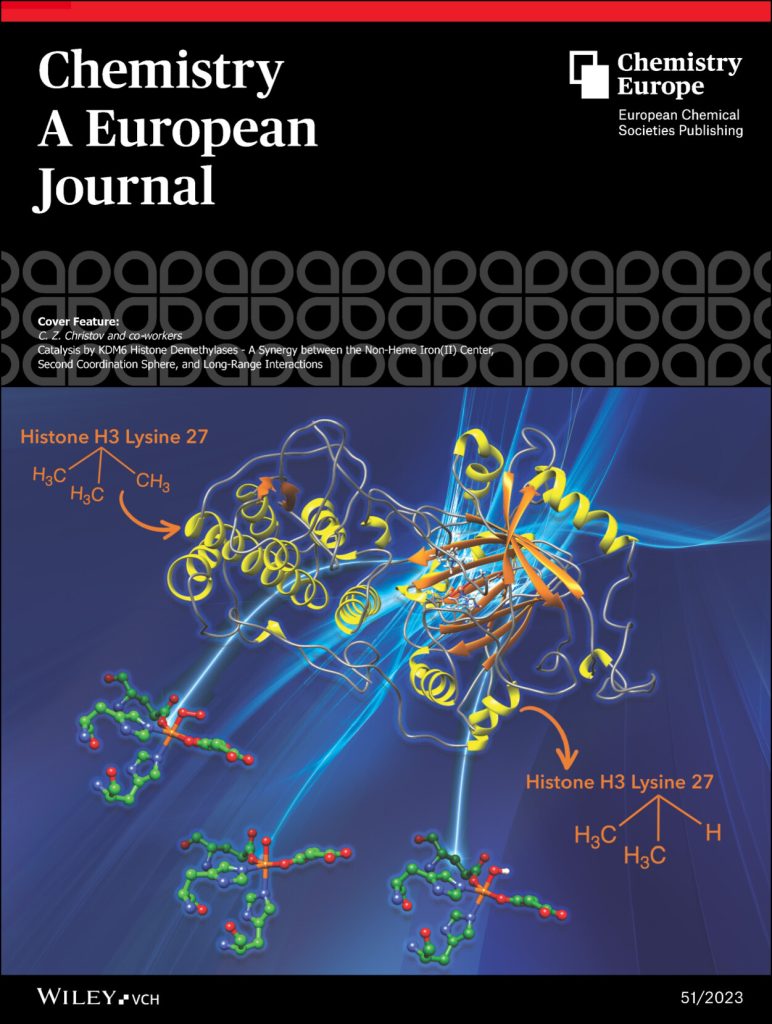
- Rifayee, S. B. J. S.; Chaturvedi, S. S.; Warner, C.; Wildey, J.; White, W.; Thompson, M.; Schofield, C. J.; Christov, C. Z. Catalysis by KDM6 Histone Demethylases – A Synergy between the Non-Heme Iron(II) Center, Second Coordination Sphere, and Long-Range Interactions. Chemistry – A European Journal, 2023. https://doi.org/10.1002/chem.202301305.
- Chaturvedi, S. S.; Rifayee, S. B. J. S.; Ramanan, R.; Rankin, J. A.; Hu, J.; Hausinger, R. P.; Christov, C. Can an External Electric Field Switch between Ethylene Formation and L-Arginine Hydroxylation in the Ethylene Forming Enzyme? Phys. Chem. Chem. Phys. 2023. https://doi.org/10.1039/D3CP01899G.
- Hausinger, R. P.; Rifayee, S. B. J. S.; Thomas, M. G.; Chatterjee, S.; Hu, J.; Christov, C. Z. Biological Formation of Ethylene. RSC Chem. Biol. 2023, 4 (9), 635–646. https://doi.org/10.1039/D3CB00066D.
- Chaturvedi, S. S.; Thomas, M. G.; Rifayee, S. B. J. S.; White, W.; Wildey, J.; Warner, C.; Schofield, C. J.; Hu, J.; Hausinger, R. P.; Karabencheva‐Christova, T. G.; Christov, C. Z. Dioxygen Binding Is Controlled by the Protein Environment in Non‐heme Fe II and 2‐Oxoglutarate Oxygenases: A Study on Histone Demethylase PHF8 and an Ethylene‐Forming Enzyme. Chemistry A European J 2023. https://doi.org/10.1002/chem.202300138.
2022
- Sosa Alfaro, V.; Waheed, S. O.; Palomino, H.; Knorrscheidt, A.; Weissenborn, M.; Christov, C. Z.; Lehnert, N. YfeX – A New Platform for Carbene Transferase Development with High Intrinsic Reactivity. Chemistry 2022, 28 (65), e202201474. https://doi.org/10.1002/chem.202201474.
- Chaturvedi, S. S.; Jaber Sathik Rifayee, S. B.; Waheed, S. O.; Wildey, J.; Warner, C.; Schofield, C. J.; Karabencheva-Christova, T. G.; Christov, C. Z. Can Second Coordination Sphere and Long-Range Interactions Modulate Hydrogen Atom Transfer in a Non-Heme Fe(II)-Dependent Histone Demethylase? JACS Au 2022, 2 (9), 2169–2186. https://doi.org/10.1021/jacsau.2c00345.
- Waheed SO, Varghese A, Chaturvedi SS, Karabencheva-Christova TG, Christov CZ. How Human TET2 Enzyme Catalyzes the Oxidation of Unnatural Cytosine Modifications in Double-Stranded DNA. ACS Catal. 2022 April 19;12:5327-5344. doi: 10.1021/acscatal.2c00024.
2021
- Ramanan R, Waheed SO, Schofield CJ, Christov CZ. What is the Catalytic Mechanism of Enzymatic Histone N-Methyl Arginine Demethylation and Can It be Influenced by an External Electric Field? Chem. Eur. J. May 14. doi: 10.1002/chem.202101174.
- Waheed SO, Chaturvedi SS, Karabencheva-Christova TG, Christov CZ. Catalytic Mechanism of Human Ten-Eleven Translocation-2 (TET2) Enzyme: Effects of Conformational Changes, Electric Field, and Mutations. ACS Catal. 2020 Mar 14;11(7):3877-3890. doi: 10.1021/acscatal.0c05034.
- Chaturvedi SS, Ramanan R, Hu J, Hausinger RP, Christov CZ. Atomic and Electronic Structure Determinants Distinguish between Ethylene Formation and l-Arginine Hydroxylation Reaction Mechanisms in the Ethylene-Forming Enzyme. ACS Catal. 2021 Jan 19;11:1578–1592. doi: 10.1021/acscatal.0c03349.
2020
- Ramanan R, Chaturvedi SS, Lehnert N, Schofield CJ, Karabencheva-Christova TG, Christov CZ. Catalysis by the JmjC histone demethylase KDM4A integrates substrate dynamics, correlated motions and molecular orbital control. Chem. Sci. 2020 Sept 4;11:9950-9961. doi: 10.1039/D0SC03713C
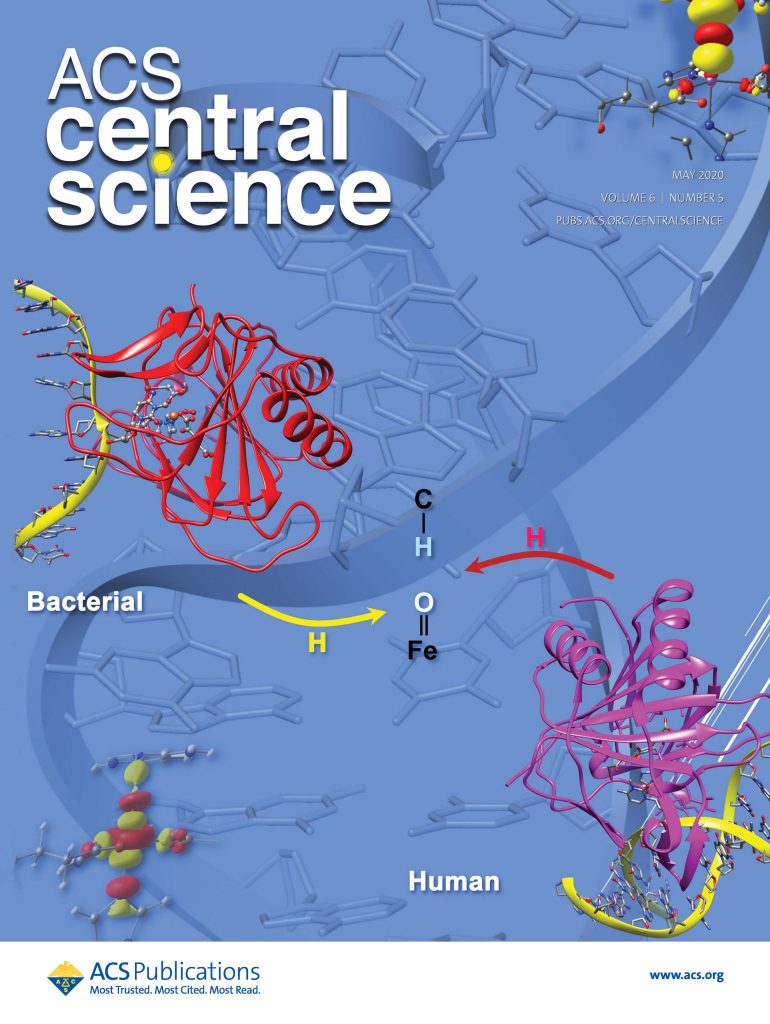
- Waheed SO, Ramanan R, Chaturvedi SS, Lehnert N, Schofield CJ, Christov CZ, Karabencheva-Christova TG. Role of Structural Dynamics in Selectivity and Mechanism of Non-heme Fe(II) and 2-Oxoglutarate-Dependent Oxygenases Involved in DNA Repair. ACS Cent Sci. 2020 May 27;6(5):795-814. doi: 10.1021/acscentsci.0c00312.
- Chaturvedi SS, Ramanan R, Lehnert N, Schofield CJ, Karabencheva-Christova TG, Christov CZ. Catalysis by the Non-Heme Iron(II) Histone Demethylase PHF8 Involves Iron Center Rearrangement and Conformational Modulation of Substrate Orientation. ACS Catal. 2020 Jan 17;10(2):1195-1209. doi: 10.1021/acscatal.9b04907.
2019
- Chaturvedi SS, Ramanan R, Waheed SO, Ainsley J, Evison M, Ames JM, Schofield CJ, Karabencheva-Christova TG, Christov CZ. Conformational Dynamics Underlies Different Functions of Human KDM7 Histone Demethylases. Chemistry. 2019 Apr 11;25(21):5422-5426. doi: 10.1002/chem.201900492.
- Waheed SO , Ramanan R , Chaturvedi SS , Ainsley J , Evison M , Ames JM , Schofield CJ , Christov CZ , Karabencheva-Christova TG . Conformational flexibility influences structure-function relationships in nucleic acid N-methyl demethylases. Org Biomol Chem. 2019 Feb 20;17(8):2223-2231. doi: 10.1039/c9ob00162j.
- Chaturvedi SS, Ramanan R, Waheed SO, Karabencheva-Christova TG, Christov CZ. Structure-function relationships in KDM7 histone demethylases. Adv Protein Chem Struct Biol. 2019;117:113-125. doi: 10.1016/bs.apcsb.2019.08.005.
2018
- Ainsley J , Chaturvedi SS , Karabencheva-Christova TG , Tanasova M , Christov CZ . Integrating molecular probes and molecular dynamics to reveal binding modes of GLUT5 activatory and inhibitory ligands. Chem Commun (Camb). 2018 Aug 30;54(71):9917-9920. doi: 10.1039/c8cc04843f.
- Ainsley J, Mulholland AJ, Black GW, Sparagano O, Christov CZ, Karabencheva-Christova TG. Structural Insights from Molecular Dynamics Simulations of Tryptophan 7-Halogenase and Tryptophan 5-Halogenase. ACS Omega. 2018 May 2;3(5):4847-4859. doi: 10.1021/acsomega.8b00385.
- Karabencheva-Christova TG, Christov CZ. Preface. Adv Protein Chem Struct Biol. 2018;113:ix. doi: 10.1016/S1876-1623(18)30054-3.
- Ainsley J, Lodola A, Mulholland AJ, Christov CZ, Karabencheva-Christova TG. Combined Quantum Mechanics and Molecular Mechanics Studies of Enzymatic Reaction Mechanisms. Adv Protein Chem Struct Biol. 2018;113:1-32. doi: 10.1016/bs.apcsb.2018.07.001.
2017
- Karabencheva-Christova TG, Torras J, Mulholland AJ, Lodola A, Christov CZ. Mechanistic Insights into the Reaction of Chlorination of Tryptophan Catalyzed by Tryptophan 7-Halogenase. Sci Rep. 2017 Dec 12;7(1):17395. doi: 10.1038/s41598-017-17789-x.
- Karabencheva-Christova TG, Christov CZ, Fields GB. Conformational Dynamics of Matrix Metalloproteinase-1·Triple-Helical Peptide Complexes. J Phys Chem B. 2018 May 31;122(21):5316-5326. doi: 10.1021/acs.jpcb.7b09771.
- Rodriguez MC, Yongye AB, Cudic M, Martinez Mayorga K, Liu E, Mueller BM, Ainsley J, Karabencheva-Christova T, Christov CZ, Cudic M, Cudic P. Targeting cancer-specific glycans by cyclic peptide lectinomimics. Amino Acids. 2017 Nov;49(11):1867-1883. doi: 10.1007/s00726-017-2485-3.
- Karabencheva-Christova TG, Christov CZ, Fields GB. Collagenolytic Matrix Metalloproteinase Structure-Function Relationships: Insights From Molecular Dynamics Studies. Adv Protein Chem Struct Biol. 2017;109:1-24. doi: 10.1016/bs.apcsb.2017.04.001.
2016
- Drummen GP, Christov CZ. Editorial: Analysis of the Interaction of Dp44mT with Human Serum Albumin and Calf Thymus DNA Using Molecular Docking and Spectroscopic Techniques. Int J Mol Sci. 2016 Nov 16;17(11):1914. doi: 10.3390/ijms17111914.
- Singh W, Fields GB, Christov CZ, Karabencheva-Christova TG. Effects of Mutations on Structure-Function Relationships of Matrix Metalloproteinase-1. Int J Mol Sci. 2016 Oct 14;17(10):1727. doi: 10.3390/ijms17101727.
- Singh W, Karabencheva-Christova TG, Black GW, Ainsley J, Dover L, Christov CZ. Conformational Dynamics, Ligand Binding and Effects of Mutations in NirE an S-Adenosyl-L-Methionine Dependent Methyltransferase. Sci Rep. 2016 Jan 29;6:20107. doi: 10.1038/srep20107.
- Christov CZ. Preface. Adv Protein Chem Struct Biol. 2016;105:ix. doi: 10.1016/S1876-1623(16)30040-2.
- Singh W, Fields GB, Christov CZ, Karabencheva-Christova TG. Importance of the Linker Region in Matrix Metalloproteinase-1 Domain Interactions. RSC Adv. 2016 Jan 1;6(28):23223-23232. doi: 10.1039/C6RA03033E.
2014
- Karabencheva TG, Lee CC, Black GW, Donev R, Christov CZ. How does conformational flexibility influence key structural features involved in activation of anaplastic lymphoma kinase? Mol Biosyst. 2014 Jun;10(6):1490-5. doi: 10.1039/c4mb00141a.
- Christov CZ. Metal-containing enzymes. Preface. Adv Protein Chem Struct Biol. 2014;97:ix. doi: 10.1016/S1876-1623(14)00055-8.
2013
- Christov CZ, Lodola A, Karabencheva-Christova TG, Wan S, Coveney PV, Mulholland AJ. Conformational effects on the pro-S hydrogen abstraction reaction in cyclooxygenase-1: an integrated QM/MM and MD study. Biophys J. 2013 Mar 5;104(5):L5-7. doi: 10.1016/j.bpj.2013.01.040.
- Christov CZ. Introduction: Biomolecular spectroscopy: advances from integrating experiments and theory. Adv Protein Chem Struct Biol. 2013;93:xi-xii. doi: 10.1016/B978-0-12-416596-0.09986-3.
- Karabencheva-Christova TG, Carlsson U, Balali-Mood K, Black GW, Christov CZ. Conformational effects on the circular dichroism of Human Carbonic Anhydrase II: a multilevel computational study. PLoS One. 2013;8(2):e56874. doi: 10.1371/journal.pone.0056874.
2011
- C. Christov, P. González-Bulnes, F. Malhaire, T. Karabencheva, C. Goudet, J.-P. Pin, A. Llebaria, and J. Giraldo. Integrated Synthetic, Pharmacological and Computational Investigation of Cis-2-(3,5-dichlorophenylcarbamoyl) Cyclohexane Carboxylic Acid Enantiomers as Positive Allosteric Modulators of Metabotropic Glutamate Receptor Subtype 4. ChemMedChem. 2011;6(1):131-140. doi: 10.1002/cmdc.201000378.
2010
- T. Karabencheva, R. Donev, Kia Balali-Mood, and C. Christov. Individual Contributions of the Aromatic Chromophores to the Near-UV Circular Dichroism in Class A β-lactamases: A Comparative Computational Analysis. Biophysical Chemistry, 2010;151(1-2):39-45. doi: 10.1016/j.bpc.2010.05.003.
- C. Christov and T. Karabencheva. Computational Insight in Protein Circular Dichroism: Detailed Analysis of Contributions of Individual Chromophores in TEM-1 ß-Lactamase. Theoretical Chemistry Accounts, 2010;128:25-37. doi: 10.1007/s00214-010-0744-4.
2008
- C. Christov, T. Karabencheva, and A. Lodola. Aromatic Interactions and Rotational Strengths within Protein Environment: An Electronic Structural Study on β-lactamases From Class A. Chemical Physics Letters, 2008;456:89-95. doi: 10.1016/j.cplett.2008.03.012.
- Christov CZ, Karabencheva TG, Lodola A. Relationship between chiroptical properties, structural changes and interactions in enzymes: a computational study on beta-lactamases from class A. Comput Biol Chem. 2008 Jun;32(3):167-75. doi: 10.1016/j.compbiolchem.2008.02.003.
- A. Lodola, M. Mol, S. Rivara, C. Christov, G. Tarzia, D. Piomelli and A. Mulholland (2008) Identification of productive inhibitor binding orientation in fatty acid amide hydrolase (FAAH) by QM/MM mechanistic modelling. Chemical Communications, 214 – 216; DOI: 10.1039/b714136j.
2006
- A Soriano, R. Castillo, C. Christov, J. Andres, V. Moliner and I. Tunon (2006) Catalysis in Glycine N-Methyltransferase: Testing Electrostatic Stabilization and Compression Hypothesis, Biochemistry, 45, 14917 -14925. doi: 10.1021/bi061319k.
2005
- C. Christov, F Tielens and M. Mirazchiiski (2005) Modeling study of the influences of the aromatic transitions and the electrostatic environment on the far-UV rotational strengths in TEM-1 β- lactamase. J. Molecular Modeling, 12, 411-416. doi: 10.1007/s00894-005-0061-3.
2004
- C. Christov and T. Karabencheva (2004) Mechanisms of generation of rotational strengths of Class-A β-lactamase from Escherichia coli (TEM-1) part I: theoretical analysis of the influence of conformational changes in the near UV, Chemical Physics Letters, 396, 282-287. doi: 10.1016/j.cplett.2004.08.035.
- C. Christov, A. Kantardjev, T. Karabencheva, and F. Tielens (2004) Mechanisms of generation of rotational strengths of Class-A β-lactamase from Escherichia coli (TEM-1) part II: theoretical study of the effects of the electrostatic interactions in the near-UV, Chemical Physics Letters, 400, 524-530. doi: 10.1016/j.cplett.2004.11.008.
- T. Karabencheva and C. Christov (2004) Comparative Theoretical Study of the Mechanisms of Generation of Rotational Strengths in the Near-UV in β-Lactamases from class A Chemical Physics Letters,398, 511-516. doi: 10.1016/j.cplett.2004.09.099.
- C. Christov, D. Yanev, A. Shosheva and B. Atanasov (2004) pH-dependent quenching of the fluorescence of tryptophan residues of TEM-1 β-lactamase: combined computational and experimental study. Zeitschrift fur Naturforschung, 59c, 11/12, 824-827. doi: 10.1515/znc-2004-11-1210.
2001
- C. Christov, S. Gabriel, B Atanasov, and J. Fleischhauer (2001) Calculation of CD spectrum of Class-A β-lactamase from Escherichia coli (TEM-1). Zeitschrift fur Naturforschung, 56a, 757-760. doi: 10.1515/zna-2001-1111.