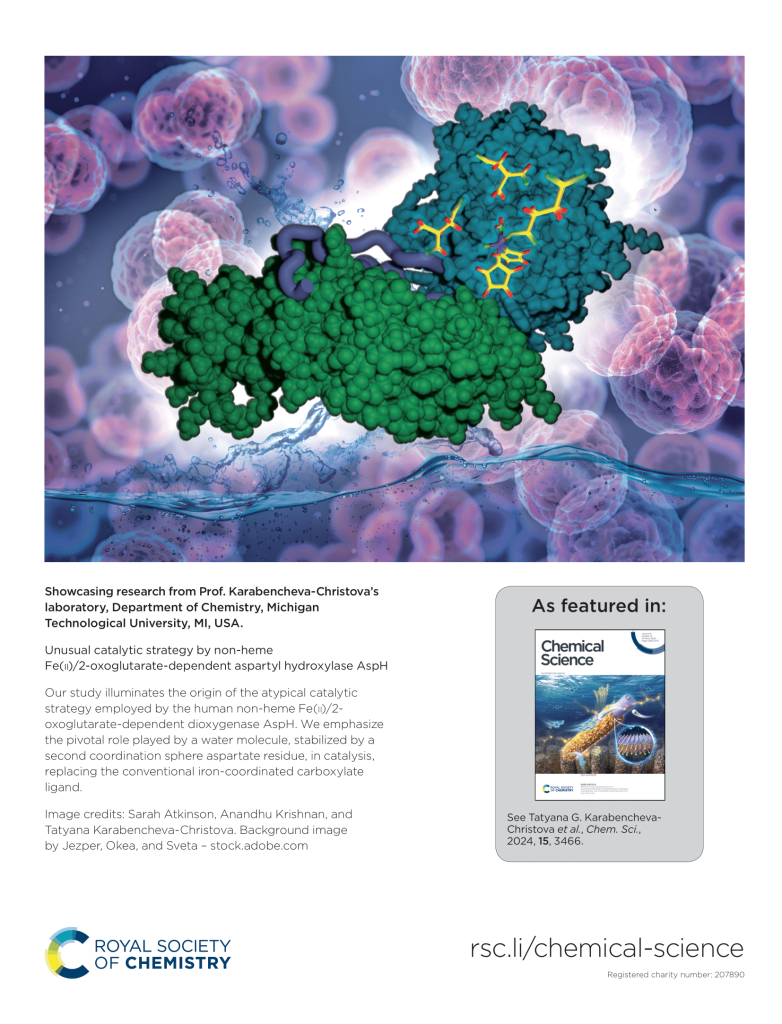
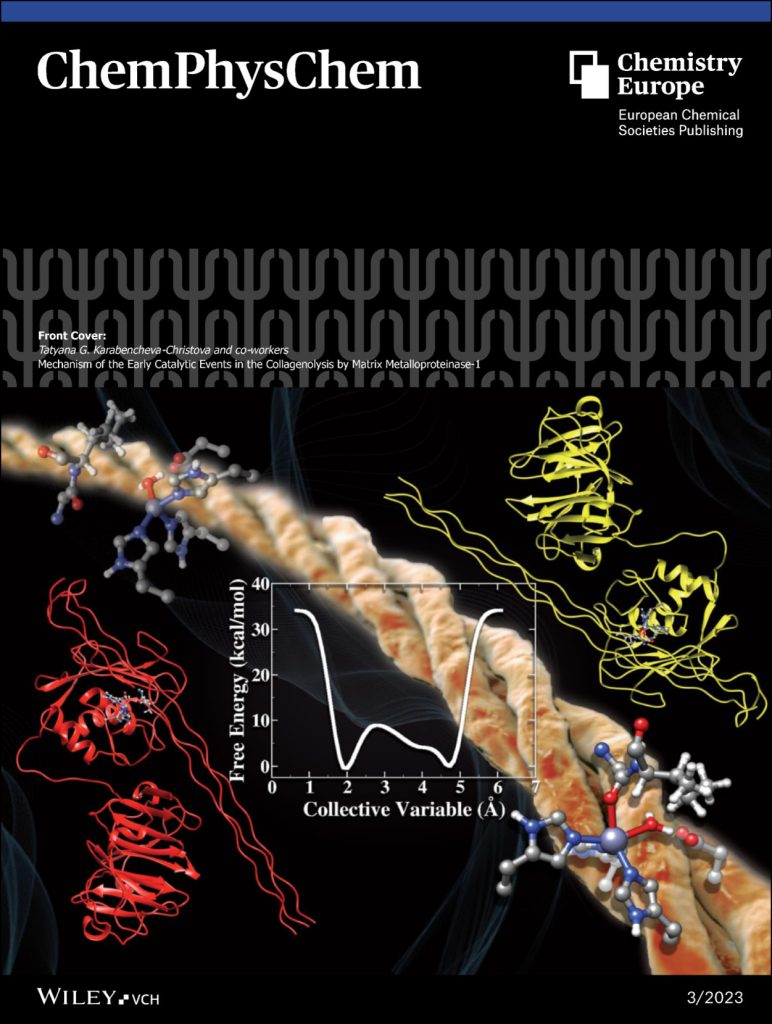
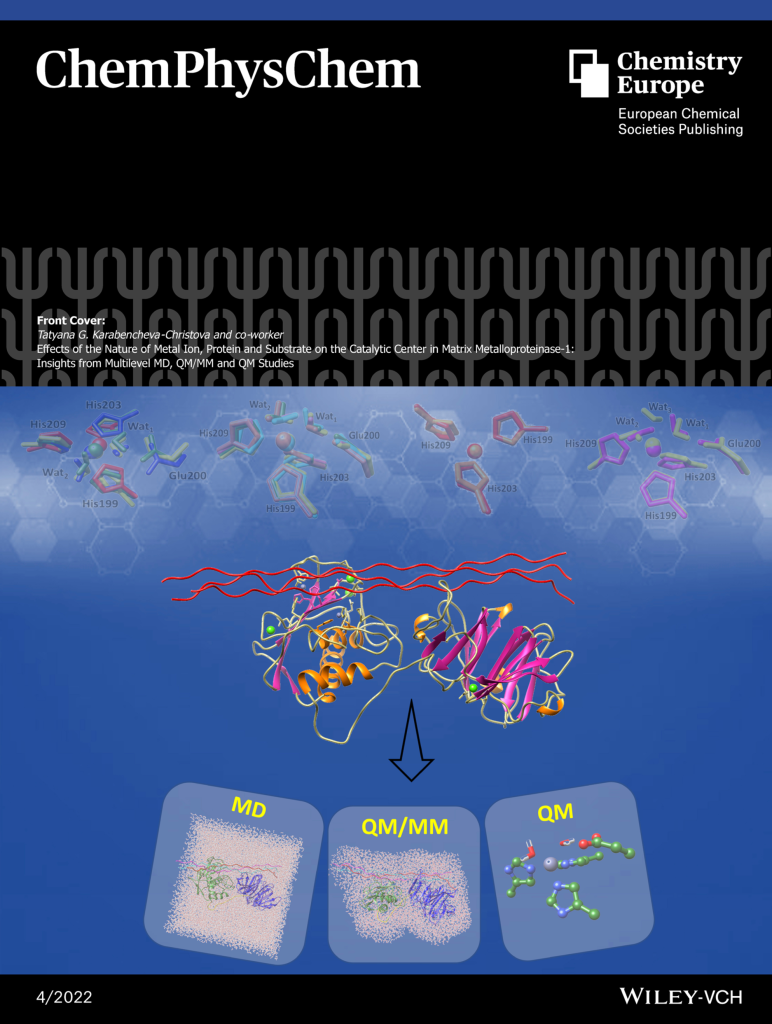
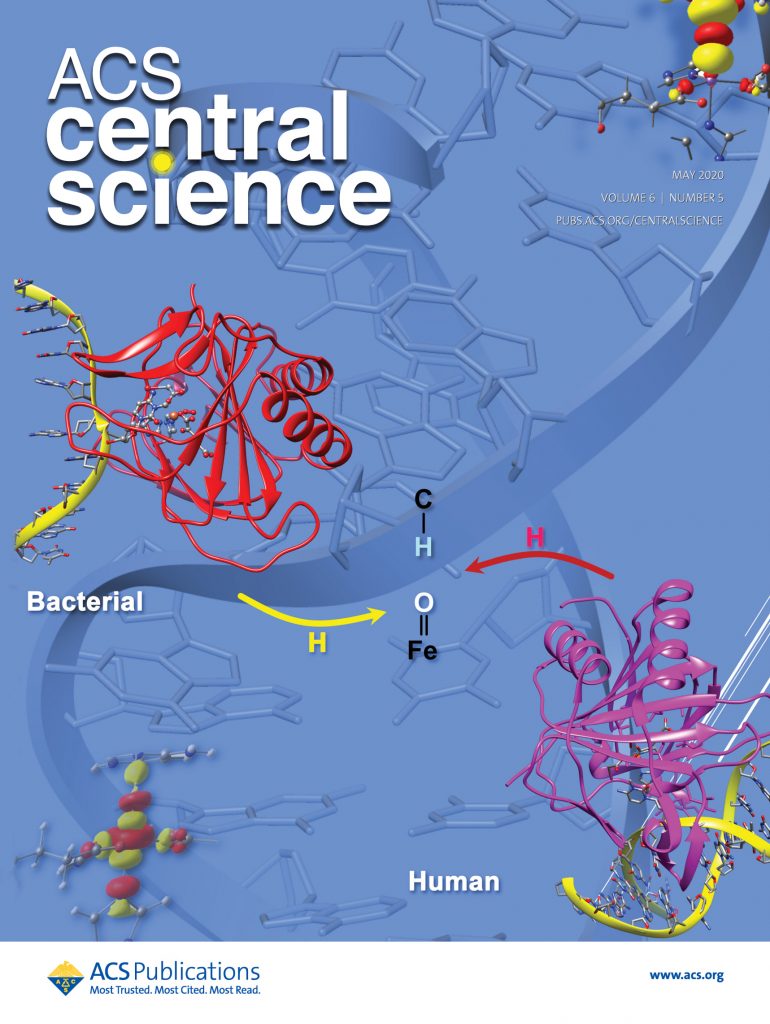
The research in our group focuses on computational studies of metalloenzymes. More than 30% of all human enzymes with essential physiological activity require metal ions. Metalloenzymes play universal functions, making them central in many human diseases and critical therapeutic targets in developing inhibitors and drugs. We provide novel insights into metalloenzymes catalytic strategy by employing hybrid quantum mechanics/molecular mechanics (QM/MM) methods and molecular dynamics (MD) simulations.